MethScope-Tutorial
MethScope-Tutorial.Rmd
library(MethScope)Overview
This tutorial demonstrates a standard MethScope workflow:
- Generate MRMP embeddings from
.cginput - Perform cell-type annotation with a pre-trained model
- Train a custom model
- Visualize prediction quality
- Run deconvolution and unsupervised clustering
Data and preprocessing resources:
- Example/reference data: https://github.com/zhou-lab/methscope_data
- Generate
.cginput files: https://zhou-lab.github.io/YAME/ - Interpret MRMP patterns: https://www.bioconductor.org/packages/release/bioc/html/knowYourCG.html
1) Generate cell/pixel/sample MRMP embeddings
Use GenerateInput() to transform a query
.cg file into a sample-by-MRMP matrix.
# Paths to your .cg and .cm files
example_file <- "/home/fuh1/cell_type/Tool/MethScope/inst/extdata/example.cg"
reference_pattern <- "/home/fuh1/cell_type/Tool/MethScope/inst/extdata/Liu2021_MouseBrain.cm"
input_pattern <- GenerateInput(example_file, reference_pattern)Example output (head(input_pattern[, 1:9])):
| Sample | P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 | P9 |
|---|---|---|---|---|---|---|---|---|---|
| GSM4012028 | 0.935 | 0.016 | 0.902 | 0.899 | 0.881 | 0.898 | 0.900 | 0.883 | 0.850 |
| GSM4014036 | 0.941 | 0.021 | 0.911 | 0.904 | 0.888 | 0.911 | 0.920 | 0.901 | 0.867 |
| GSM4014043 | 0.955 | 0.019 | 0.931 | 0.926 | 0.916 | 0.930 | 0.918 | 0.932 | 0.897 |
| GSM4014056 | 0.954 | 0.023 | 0.940 | 0.934 | 0.928 | 0.940 | 0.912 | 0.932 | 0.918 |
| GSM4014298 | 0.947 | 0.016 | 0.926 | 0.922 | 0.909 | 0.923 | 0.863 | 0.918 | 0.894 |
| GSM4016157 | 0.929 | 0.016 | 0.887 | 0.881 | 0.859 | 0.880 | 0.904 | 0.882 | 0.825 |
For really large datasets, please consider split .cg
files and process in parallel: https://zhou-lab.github.io/YAME/docs/subcommands/YAME_subset.html
2) Perform cell-type annotation
Use a built-in pre-trained model for fast annotation. Available built-in examples include:
MethScope:::Liu2021_MouseBrain_P1000MethScope:::Zhou2025_HumanAtlas_P1000
prediction_result <- PredictCellType(
MethScope:::Liu2021_MouseBrain_P1000,
input_pattern
)3) Train a custom classification model
You can train your own XGBoost model using known labels.
cell_type_label must align with rows of
input_pattern.
trained_model <- Input_training(
input_pattern,
cell_type_label
)You can also tune hyperparameters automatically or provide your own list of xgb_parameters:
trained_model_cv <- Input_training(
input_pattern,
cell_type_label,
cross_validation = TRUE
)4) Visualize prediction results
umap_plot <- PlotUMAP(input_pattern, prediction_result)
# cell_type_label is the true label vector
PlotConfusion(prediction_result, cell_type_label)
PlotF1(prediction_result, cell_type_label)
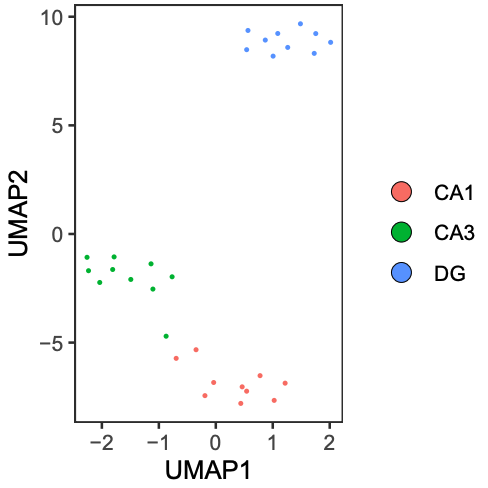
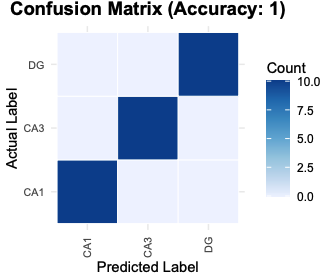
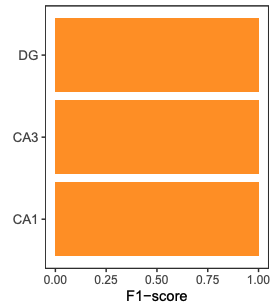
5) Cell-type deconvolution
Estimate cell-type proportions using nnls_deconv(). Some
reference matrix examples are available in: https://github.com/zhou-lab/methscope_data
reference_pattern <- "Liu2021_MouseBrain.cm"
reference_input <- readRDS("2021Liu_reference_pattern.rds")
cell_proportion <- nnls_deconv(reference_input, input_pattern)6) Unsupervised clustering (Seurat example)
After generating input_pattern, you can cluster with
standard single-cell pipelines.
library(Seurat)
Pattern.obj <- CreateSeuratObject(counts = t(input_pattern), assay = "DNAm")
VariableFeatures(Pattern.obj) <- rownames(Pattern.obj[['DNAm']])
DefaultAssay(Pattern.obj) <- "DNAm"
Pattern.obj <- NormalizeData(Pattern.obj, assay = "DNAm", verbose = FALSE)
Pattern.obj <- ScaleData(Pattern.obj, assay = "DNAm", verbose = FALSE)
# You can also directly use the initial counts matrix
Pattern.obj@assays$DNAm@layers$scale.data <- as.matrix(Pattern.obj@assays$DNAm@layers$counts)
Pattern.obj <- RunPCA(Pattern.obj, assay = "DNAm", reduction.name = "mpca", verbose = FALSE)
Pattern.obj <- FindNeighbors(Pattern.obj, reduction = "mpca", dims = 1:30)
Pattern.obj <- FindClusters(Pattern.obj, verbose = FALSE, resolution = 0.7)
Pattern.obj <- RunUMAP(Pattern.obj, reduction = "mpca", reduction.name = "meth.umap", dims = 1:30)7) Upscaling Network (10k-CpG block example)
This section describes a compact workflow to upscale MRMP embeddings to CpG-level methylation predictions within a genomic block.
7.1 Prepare model input (x) and target
(y)
x is the MRMP embedding matrix (samples x
features).y is the CpG-level ground truth matrix for the same samples
(samples x CpGs).
# create masked replicates (masked CpGs encoded as 2)
# this generate 100 replicates per original sample, each of the 10 output files contains 10 replicates per sample
parallel -j 24 'yame dsample -p {} -s {} -r 10 -N 29000 -o example_rep{}.cg example.cg' ::: {1..10}
# summarize MRMP features per replicate
parallel -j 24 'yame summary -m patterns.cm example_rep{}.cg | cut -f2,4,10 | doawk | sort -k1,1 -k2,2n | bedtools groupby -g 1 -c 3 -o collapse > example_rep{}_mrmp.txt' ::: {1..10}
cat example_rep*_mrmp.txt > input_pattern.txt
# whole-genome .cg file can be divided into 10k-CpG blocks, and then CpG-level truth from .cg
parallel -j 72 'yame rowsub -I {}_10000 example.cg | yame unpack -a - >truth/example_{}.txt' ::: {0..2940}
x <- read.table("input_pattern.txt", header = FALSE)
y <- read.table("example_0.txt", header = FALSE)
y <- t(y) # samples x CpGs7.2 Train decoder (Python)
import torch
import torch.nn as nn
class BottleneckDecoder(nn.Module):
def __init__(self, input_dim=num_of_patterns, hidden_dim=512, output_dim=10000):
super().__init__()
self.net = nn.Sequential(
nn.Linear(input_dim, hidden_dim),
nn.ReLU(),
nn.BatchNorm1d(hidden_dim),
nn.Linear(hidden_dim, output_dim)
)
def forward(self, x):
return self.net(x)Recommended trainning setup:
- Loss: masked BCEWithLogitsLoss (ignore entries where label is 2)
- Optimizer: Adam (lr = 1e-3, weight_decay = 1e-5)
- Scheduler: ReduceLROnPlateau
- Model selection: run 3 epoches and save checkpoint with best validation
7.3 Evaluate
After training:
1. Run inference on the test set.
2. Apply sigmoid to logits to obtain probabilities.
3. Compute metrics on valid positions only (Accuracy, MAE, AUPRC).
4. Save predicted probabilities to predictions.csv for downstream R
visualization.
Notes
- For whole-genome analysis, perform block-wise upscaling in parallel to
improve computational efficiency.
Related resources
- MRMP reference tutorial: https://zhou-lab.github.io/MethScope/articles/MethScope-MRMP.html
- Full package site: https://zhou-lab.github.io/MethScope/